As promised at the end of my post on polar and non-polar molecules, here’s a basic concepts post on intermolecular forces. Intermolecular forces are the forces between molecules, whereas intramolecular forces are those within molecules. (The bonds that hold the atoms in a molecule together are intramolecular forces.)
A quick note before we jump in: When chemical educators are explaining intermolecular forces, they almost always use examples of intermolecular attractions. You could just as easily talk about intermolecular repulsions (which are forces too, after all), but it’s the attractions that result in more stable arrangements of bunches of molecules — in other words, intermolecular attractions end up doing lots of useful explanatory work.
One big thing intermolecular forces help to explain is the phase of matter a compound assumes at room temperature. This is because liquids and solids have molecules associated with each other by way of intermolecular forces, while gases have molecules zipping around as if they’re the only molecule in the world, paying no attention to the other molecules that may be around. In the presence of intermolecular attractions, those molecules would be paying attention to each other.
There are three key ideas behind intermolecular forces:
- Oppositely charged bits of molecules attract each other (while bits of molecules of the same charge repel each other).
- Charges of greater magnitude make for stronger attractive forces.
- Charged bits of molecules exert stronger attractive forces when they’re closer to each other than when they’re far away from each other.
These give rise to some different sorts of intermolecular forces. I’ll run through them and then give you the low-down on how you tell which intermolecular force is the most important one for a particular compound.
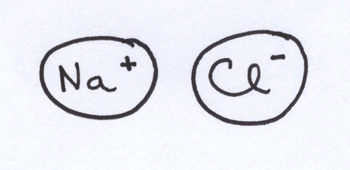 Ionic bonding. Let me acknowledge upfront that there is some disagreement about whether ionic bonding is more properly an intermolecular force or an intramolecular force — it’s what holds together large quantities of sodium chloride, but it’s also what holds together the singe Na+ and Cl– ions that you’d be inclined to identify as a molecule of NaCl.
Ionic bonding. Let me acknowledge upfront that there is some disagreement about whether ionic bonding is more properly an intermolecular force or an intramolecular force — it’s what holds together large quantities of sodium chloride, but it’s also what holds together the singe Na+ and Cl– ions that you’d be inclined to identify as a molecule of NaCl.
The proper classification of ionic bonding doesn’t keep me up at nights. And, I’m inclined to think it’s useful to take ionic bonding as an extreme case of what we’ll see in the other kinds of forces that clearly ought to be counted as intermolecular forces.
In cases of ionic bonding, ions with full positive charges and full negative charges hang out together due to electrostatic attractions between the two charged ions. Na+ and Cl– each have charges of the same magnitude (+1 and -1), but there are ions with charges of greater magnitude, and those can potentially give you stronger attractions between the ions. The other factor that makes a difference here is the size of the ion — a +1 charge on a very small ion has more attractive bang for its buck than a +3 charge spread over a very big ion.
In any compound where the atoms form ionic bonds rather than covalent bonds, ionic bonding is going to be the most important intermolecular force, simply because the electrostatic attractions between fully charged ions are extremely strong (as per #2 in the list of big ideas above).
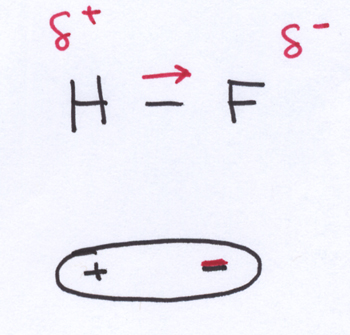 Dipole-dipole attractions. For polar molecules, which have discernible ends with partial positive charges and partial negative charges, intermolecular attractions between the positive end of one molecule and the negative end of another molecule (and vice versa) are important. The dipole-dipole attractions are not as strong as ionic bonding because the partial charges are smaller in magnitude than the full charges on ions. Still, in situations where the polar molecules are close enough to each other to notice each other’s partially charged ends, you can develop attractions that hold molecules together with each other pretty well.
Dipole-dipole attractions. For polar molecules, which have discernible ends with partial positive charges and partial negative charges, intermolecular attractions between the positive end of one molecule and the negative end of another molecule (and vice versa) are important. The dipole-dipole attractions are not as strong as ionic bonding because the partial charges are smaller in magnitude than the full charges on ions. Still, in situations where the polar molecules are close enough to each other to notice each other’s partially charged ends, you can develop attractions that hold molecules together with each other pretty well.
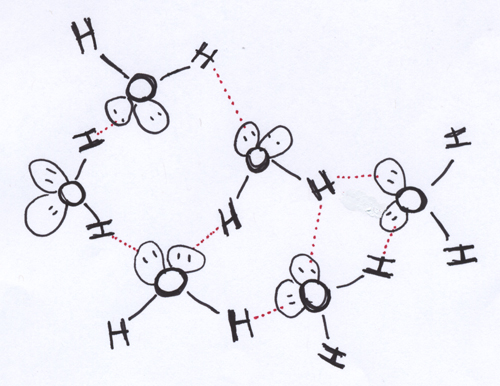
Although it is sometimes treated as if it were an entirely separate sort of intermolecular force, hydrogen bonding is basically a really strong dipole-dipole attraction. Hydrogen bonding is seen in polar molecules with bonds between H (because it’s hydrogen bonding) and N, O, or F (but probably not Cl — see the comment below). Notice that you need a polar molecule to get hydrogen bonding (since it’s really strong dipole-dipole attraction; a non-polar molecule doesn’t have the necessary positive and negative ends).
What’s so special about these atoms? N, O, and F all have a pretty high ratio of electronegativity to atomic radius. In a tug of war with H over bond electrons, these atoms pull a good bit of the electron density their direction. And, that electron density is concentrated close to a pretty small nuclear center. What this means is that the partial negative charge on the F in HF, or on the O in H2O, is coming closer to a full negative charge than does the partial negative charge on your run-of-the-mill polar molecule. The same kind of consideration applies to the H here: it’s lost enough electron density in the bond tug of war, and it’s a small enough nucleus, that its partial positive charge is creeping up on being a full positive charge.
Again, #2 in our list of guiding ideas reminds us that the bigger the charges, the stronger the attraction. The bigger partial charges in the polar molecules with H bonded to N, O, F, or Cl make for stronger dipole-dipole attractions. So, hydrogen bonding is the über-dipole-dipole attraction. Here it is in my cartoon of the associations between water molecules:
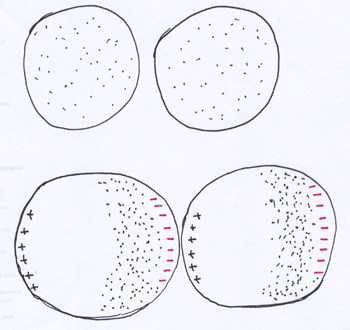 London or van der Waals forces. You’ve noticed by now that only polar molecules can avail themselves of dipole-dipole attractions (including hydrogen bonding). That’s well and good for the polar molecules, but there are oodles of compounds whose molecules are non-polar. What are they supposed to do to form liquids and solids? Because verily, there are non-polar compounds that form liquids and solids. The London or van der Waals forces are how they do it.
London or van der Waals forces. You’ve noticed by now that only polar molecules can avail themselves of dipole-dipole attractions (including hydrogen bonding). That’s well and good for the polar molecules, but there are oodles of compounds whose molecules are non-polar. What are they supposed to do to form liquids and solids? Because verily, there are non-polar compounds that form liquids and solids. The London or van der Waals forces are how they do it.
What is a molecule? It’s a bunch of atoms bonded together (as described earlier). So you have the positively charged nuclei of the molecule’s atoms hooked together with bonds (made of electrons). And, usually the nuclei have other electrons hanging out nearby that aren’t involved in the intramolecular bonds. In other words, you can think of a molecule as a structure that has a cloud of electron density around it and the positive nuclear charges buried underneath that electron cloud.
The electron cloud can — and does — shift. You don’t always have an even distribution of the electrons in the cloud. At some moments there’s more electron density on one side of the molecule than the other. At such moments, what develops is like a wannabe dipole: there’s one side of the molecule with just a hint of negative character from having more of the cloud on that side, while the other end has a bit more of its positive character from the nuclei underneath exposed by the thinner side of the electron cloud. The hint of charge on each end is much smaller than the partial positive and partial negative charges found in real polar molecules.
But, if nearby non-polar molecules get their electron clouds shifting in sync with each other, so that the hint of positive character on one molecule is nice and close to the hint of negative character on the adjacent molecule, the tiny little attractions between them can be enough to hold the molecules together into a liquid (like bromine, Br2, or carbon tetrachloride, CCl4), or even a solid (like iodine, I2). The more electrons the molecule has to throw around in the electron cloud, the more significant the London/van der Waals forces will be.
The London/van der Waals forces are the only intermolecular force available to non-polar molecules. Since polar molecules also have electron clouds, they have London/van der Waals forces too. However, they are so much weaker than dipole-dipole attractions that they are negligible for polar molecules.
With an understanding of intermolecular forces, you have a good basis for figuring out which molecules are going to “stick together” in solids and liquids, and which will be more likely to be found as gases. There’s a good chance I’ll write a post on phase changes in the next day or two.

I’d like to suggest that hydrophobic interactions deserve a mention. Perhaps a whole separate post, since they are distinguished by not being electrostatically-driven, in contrast to the ones you have discussed. In aqueous media the hydrophobic interaction often becomes the strongest of the non-covalent “weak bonds” since the high dielectric of water tends to attenuate all charge-charge interactions.
“The London/van der Waals forces are the only intermolecular force available to non-polar molecules”: – no, that is just plain wrong. You are leaving out an entire class of electrostatic interactions: permanent moments beyond the dipole. Take, for example, carbon dioxide, CO2. It has no dipole moment, but it has a very large quadrupole moment, corresponding to a charge distribution that is displaced towards the oxygens and away from carbons. This is the leading term in the intermolecular interaction energy, much more important than the London Dispersion forces. Then there are induction terms involving permanent quadrupole inducing dipole moments, etc.
It’s only at large intermolecular separations that the the electrostatic interaction is dominated by permanent dipoles. At short distances – i.e. at the separations corresponding to molecules in condensed phases – it’s better to throw away the multipole expansion and treat the molecules as extended charge distributions.
“Although it is sometimes treated as if it were an entirely separate sort of intermolecular force, hydrogen bonding is basically a really strong dipole-dipole attraction.”
Not true. Hydrogen-bonded systems have significant covalent character. Only the ionic contributions to the wavefunction can be properly said to contribute to dipole-dipole interactions.
Robert and Elia, your points are well taken, but the intent of this post is to set out the basic concepts behind intermolecular attraction. In other words, I’m setting out a simple model. Clearly, we could take more things into account (like quadrupole moments), but then the model is no longer in the realm of the basic. And, once we’re too far from basic, the concept is not necessarily all that useful to someone who can’t already break out the wavefunctions.
The simplified model approach is pretty much how we grow people’s chemical intuitions. Once those intuitions have taken root, we can double back and make the models more sophisticated. Does this mean we’re lying (or badly misleading) when we set out the simplified models? Perhaps, but even the more sophisticated models depart from the real phenomena they are used to represent. This is part of what makes them models, after all.
And, for the record, let me quote the first intro chem text I pulled off the shelf (Whitten, Gailey, and Davis, General Chemistry with Qualitative Analysis, 4th edition) to double-check my recollection of how this stuff if usually presented:
It does seem, however, that polar molecules with H-Cl bonds are enough of a “borderline” case that it might be iffy to describe their intermolecular attractions as hydrogen bonding. So I’ll fix that part of the post.
Wow! This is great. I have an essay on inter/intra molecular forces to do and this stuff helps tons. Much better than that “boring” science stuff found on wikipedia. Grade 11 chemistry is going to be a breeze this year with my new found source.
Thanks!
Great post and commentaries. I have never gotten into the finer points of chemistry. (Despite a background in solid state physics – alas, it was more electronics and material technology.) For example, before reading Robert’s comment I would have said that the following is wrong:
since another reason for dipoles, at least fixed ones, is weaker is that fields from dipoles falls as r^-3, faster than monopoles as r^-2. (And so on for moments from higher multipoles, and similarly for dispersion forces at approximately r^-6.)
But if we should treat molecules as extended charges for the range of interest (which at least sounds familiar), I guess the above is correct as well in the simple model.
Janet, it’s ok – indeed, it is essential – to simplify when making a presentation to beginners. I’ve been teaching General Chemistry for nearly 20 years now, believe me I struggle with this dilemma all of the time. It is *NOT* ok to tell students things that are manifestly false, like “The London/van der Waals forces are the only intermolecular force available to non-polar molecules.” This is not a case of simplifying a model so as to make it comprehensible – students who understand the idea of electronegativity to the degree that they can appreciate polarity in HF, HCl, or H2) can just as easily appreciate the separation of charge in CO2 – and can then understand why CO2 freezes a few tens of
degrees below zero celsius, whereas Ar (no charge separation, London forces only) or N2 (very small charge separation, electrostatic forces small and comparable to London forces) only liquify/freeze at much lower temperatures.
Torbjorn – your instincts are basically on track. It has been shown that conventional single-center multipole expansion has a radius of convergence that is larger than the characteristic radius of a molecule (cf. A. J. Stone, _The Theory of Intermolecular Forces_, Oxford, late 1990’s). Thus, when dealing with long, skinny or flat molecules in solids or liquids, it is better to use a “distributed multipole analysis” – a nonorthogonal expansion of the charge distribution around several sites on each molecule. These converge rapidly, can readily be extracted from ab-initio electronic structure calculations, and nevertheless preserve a connection to the chemist’s intuitive ideas about electronegativity and charge separation. (For example, one can describe the molecular dynamics of solid CO2 very nicely in terms of a distributed charge model in which partial negative charges a)e placed on the oxygen atoms and partial positive on the C.